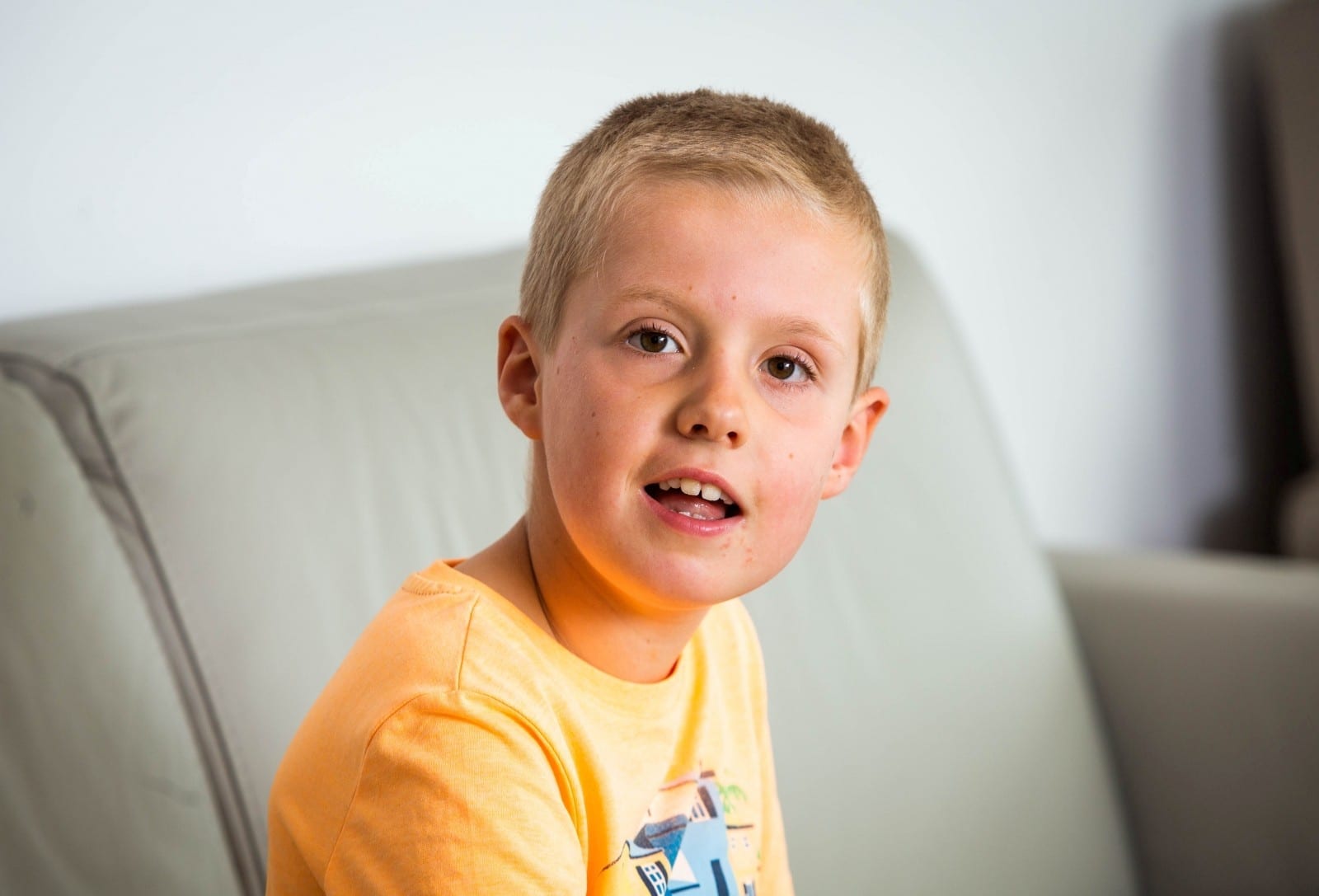
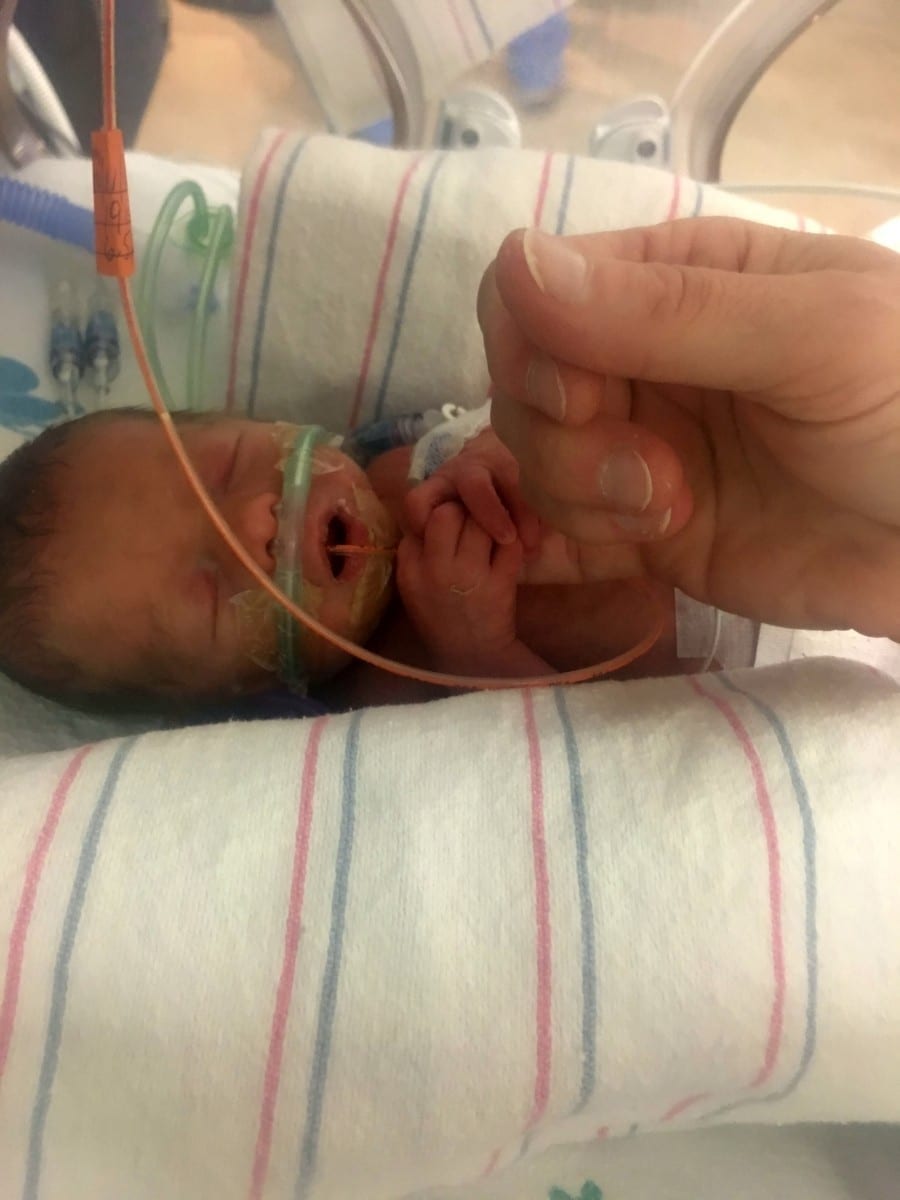
An eight-year-old with a rare genetic disorder that is slowly killing him has been forced to moves to the Netherlands – as his medicine is not available on the NHS.
Max Sewart and his dad Simon have moved to Rotterdam so he can be treated for a rare type of Batten Disease – leaving mum Ivana and younger brother Felix in Britain.
The youngster became involved in a stand-off between NHS chiefs and the manufacturer of medication to treat the rare fatal disease.

NHS bosses in England are refusing to pay the asking price for the drug, cerliponase alfa – marketed as Brinuera – even though it is available in 20 countries including Wales.
It is said to slow down the progression of Batten disease and even stabilise it in cases.
Max, from Chew Valley, Somerset, was only diagnosed with the condition a few months ago and has rapidly deteriorated even though the drug that could stop that exists.
His family set up a crowdfunder, raising £15,000, to try to fund it themselves as well as a treatment called critical enzyme replacement therapy.
But they need to raise £500,000 to fund what they see as a potential cure.
Mr Sewart has joined forces with the families of just over a dozen other children in the same position in the battle with the NHS – after their talks with drug manufacturers broke down.
Max and Simon flew out from Bristol Airport on Tuesday and took a train from Amsterdam to Rotterdam to set up home.
There they will be eligible for treatment under the Dutch health service – something which would not have been possible had they simply moved to Wales.

Simon said: “It’s an absurd situation. It’s bad enough with the diagnosis, but to have to do this nonsense too.
“I feel so badly let down by the NHS – it’s not what the service is about.
“It’s about looking after your society. Their negotiators are not humane people.
“It’s a rare disease drug, so it’s very expensive.
“We want to preserve Max in the state he’s in.
“We’ve seen his decline in the last four months.
“We’ve given up on the NHS.”
The Sewarts’ MP, Jacob Rees Mogg, has backed their campaign, and raised the case – and those of other children – in the House of Commons several times.
Last week, he did so again, demanding answers from the NHS and health ministers.
“The terrible thing is that in the month that has passed since I first raised this matter in the House, Max’s condition will have slightly deteriorated, and in every month that goes on while we are debating this, not just Max but all the children with this condition will go downhill. That is what happens with this disease,” he told MPs last week.
“Although I accept, of course, that there is a need to look at costs, when we are talking about eight-year-old children, we are not talking about a cost for people who only have weeks or months to live, but about a child who could have years of a high quality of life ahead of him.
“That must be where most of us as taxpayers think it is right to spend money and where we think that the moral case for spending money is extraordinarily strong.”
At the weekend, Mr Rees Mogg was one of hundreds who attended the village fete in East Harptree, which was this year held as a fundraiser for Max.
Organised jointly between the school and the church, a record £5,000 was raised towards the public appeal to help the family.

Max and Felix’s headteacher at the village primary school, Jane Bailey, said she was proud of the response from the village.
“We are striving to support Max, his younger brother and parents at this desperately challenging time within school,” she said.
“However, we felt we wanted to do more as a community, especially as we have heard that NHS England has refused the vital treatment that Max needs to slow down the symptoms of this degenerative and life shortening disease.
“I’m immensely proud of all our children and their families for responding so generously with their time, donations and generosity.”
An NHS England spokeswoman said: “The National Institute for Health and Care Excellence (NICE) have not recommended this drug, since British taxpayers simply cannot offer a blank cheque to this particular American pharmaceutical company BioMarin, who have sadly so far shown a complete lack of willingness to price their products fairly, as independently assessed by NICE.”
A spokeswoman for BioMarin said: “BioMarin is deeply disappointed by the NICE and NHS England decision to not approve cerliponase alfa for the treatment of CLN2 disease, this is a tragic outcome for children living in England with this debilitating disease.
“Patients with ultra-rare diseases in England face greater barriers to access effective new and innovative medicines through restrictions imposed by the changes in the NICE HST appraisal process, compared to other countries worldwide.
“It should be noted that the 11 patients already receiving treatment in England are all currently being given treatment free by BioMarin.
By doing so BioMarin is doing everything it can to help patients and their families, but the company cannot be expected to simply treat all patients for free – it is not a sustainable solution.”
She continued: “Since the decision has been made BioMarin has provided alternative proposals but NHS England has refused to discuss these with us.
“The company has reached agreements for access to Brineura across most of Europe (which includes Wales, the Netherlands, Germany, France, Hungary, Italy, Serbia and Ukraine).
“We remain strongly committed to ongoing discussions with NICE, NHS England and all relevant stakeholders in the CLN2 community to engage in meaningful discussions and negotiate a sustainable solution that will allow all eligible children access to this breakthrough therapy for all children living with the condition in England.
“As an interested party in an ongoing judicial review, BioMarin is unable to provide further information or comment at this time.”
Commonly referred to as Batten disease, the Neuronal Ceroid Lipofuscinoses (NCLs) denote several different genetic life-limiting neurodegenerative diseases that share similar features.
The NCLs are caused by abnormal genes, which are unable to produce the required proteins. As a result, the cells do not work properly and this leads to the development of symptoms associated with these diseases.
It is estimated that there are approximately 100 – 150 children, young people and adults currently living with an NCL diagnosis in the UK.
Symptoms include visual impairment, epilepsy, seizures, difficulty sleeping and the loss of mobility.
Death is inevitable and, depending on the age of onset and specific genetic diagnosis, may occur anywhere between early childhood and young adulthood.
All information taken from the Batten Disease Family Association website.